By Mackenzie Carpenter
Pittsburgh Post-Gazette.
PITTSBURGH:
She’s 35 pounds overweight and has tried everything, but when that patient goes to see George Eid, a bariatric surgeon in the Allegheny Health Network, he usually has to tell her she’s not heavy enough for weight-loss surgery.
Soon, though, there may be a new option for that overweight patient who doesn’t need to lose 100 pounds, but maybe a third of that, to jump start a weight-loss program, perhaps, or prepare for a surgical procedure.
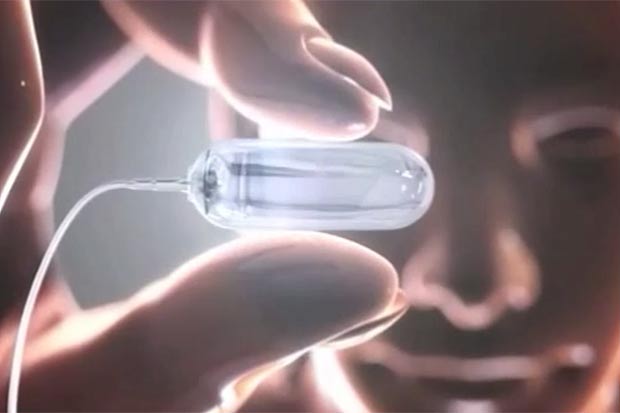
Eid, along with doctors at 15 medical centers around the country, is testing a new pill that, once swallowed, is pumped full of nitrogen gas to inflate a balloon in the stomach.
“The idea is that with the balloon taking up space, patients will feel fuller faster and eat less,” said Eid, director of AHN’s Bariatric and Metabolic Institute. “This is really a way to help people who are on their way to becoming obese but maybe aren’t there yet. If we halt weight gain early enough in the process, they may never have to get there.”
The balloon was developed by Obalon Therapeutics, a California-based company, which has a video on its website (www.BalloonTrial.com) that shows how it’s done.
The procedure, which is already approved for use in Europe, involves three balloons placed in the stomach over a period of several weeks. The first balloon is swallowed in the form of an inflatable capsule, with a tube attached. The gas is then pumped into the tube and then an X-ray determines that it has been properly placed. The tube is then removed.
After six months, the balloons are removed using a flexible tube camera that is inserted into the mouth. Then, the patient, who theoretically has lost weight, will continue for the next six months with diet and exercise counseling and, possibly, weight-loss medication.
Patients enrolled in the clinical study, Eid said, will be randomly placed into two groups: one gets the pill containing the Obalon Balloon, while the other gets a sugar pill, or “sham” device. Those in the second group will, after the initial six-month period, be offered the balloon at no cost.
Participants must be between the ages of 22 and 64; have a body mass index, or BMI, of between 30 and 40; and not have had any previous surgeries.
Eligibility for weight-loss surgery is based on one’s BMI, Eid said. “If a patient comes to me with a BMI of over 40, there’s no problem. They should get surgery since it’s the only proven and effective way to treat that level of obesity. If the BMI is between 35 and 40, it’s a gray zone, patients are eligible if they have co-morbidities, conditions such as diabetes or sleep apnea. If you come in with a 30 to 35 BMI, you are technically in what is known as Class 1 obesity, but I can’t offer you any (surgical) interventions.”
The Food and Drug Administration is expected to approve other weight-loss balloon technologies in the coming months, Eid said, adding that hundreds of federal studies show that obese patients with a BMI of 30 and above are resistant to diet and exercise. Those other techniques use saline to inflate the balloon.
“This is all aimed at addressing unmet needs in a population that still needs help,” Eid said. “I’m sure people get flak for doing this, but if it means we can eliminate diabetes through this kind of weight-loss intervention instead of giving medication, isn’t that OK?”