By Mary Divine
Pioneer Press, St. Paul, Minn.
WWR Article Summary (tl;dr) Teresa Tollefson is helping build the world’s smallest pediatric mechanical heart valve for children with heart defects.
Pioneer Press, St. Paul, Minn.
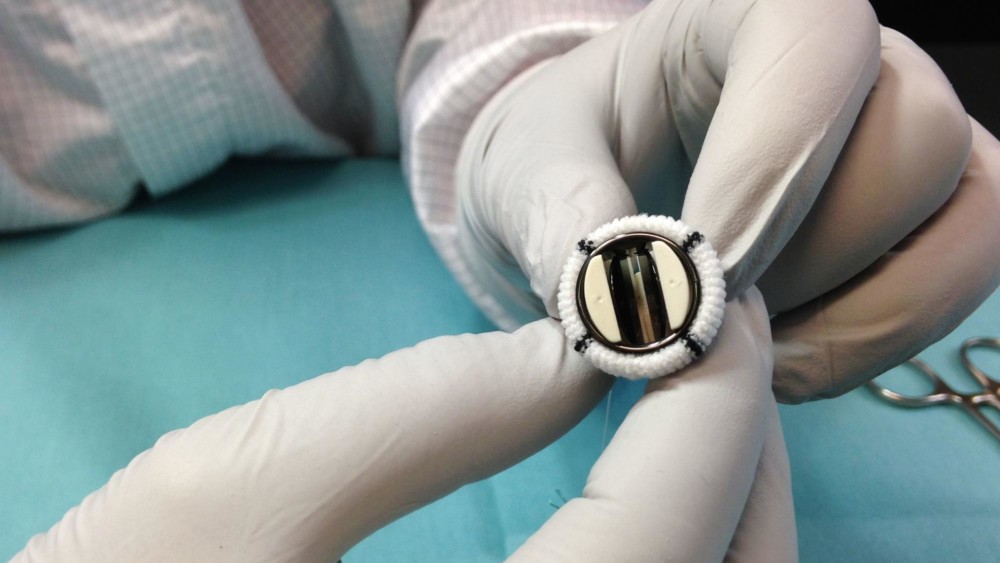
In a laboratory near Interstate 35E and Minnesota 36, Abbott Laboratories employee Teresa Tollefson peers through a microscope, dips a Q-tip in Isopropyl alcohol and gently swabs a ring the size of a dime.
First, she turns the microscope to its 7X setting. If she sees any anomalies in the pyrolytic carbon ring, she’ll bump up to 30X “for closer inspection,” she said.
Tollefson is helping build the world’s smallest pediatric mechanical heart valve for children with heart defects.
Recently approved by the U.S. Food and Drug Administration, the valve is the first deemed suitable for newborns and young infants.
It is the smallest mechanical heart valve available commercially in the world.
“It’s very gratifying, knowing that it’s a baby valve, and it could ultimately save a child’s life,” said
Tollefson, who has worked at the former St. Jude Medical manufacturing facility in Little Canada for 21 years.
“They are going to potentially have a full healthy life, and this device is going to give them that.”
The work is labor-intensive. Each 15-millimeter valve takes about two weeks to produce. Some of the parts must be hand-sewn into place.
SURGEONS’ REQUESTS
Officials at St. Jude Medical, which was acquired by Illinois-based Abbott in 2017, began making the miniature valves in 2012 at the urging of pediatric cardiac surgeons.
“We kept getting requests” ‘Boy, we could really use one of these for the smallest of the smallest children,’ so we’re taking our technology and applying it for infants,” said Rick Olson, divisional vice president of product development for Abbott’s structural heart business. “Before this, there was nothing available to them.”
Congenital heart defects affect nearly 1 percent — about 40,000 — births each year in the U.S. Of those, about one in four babies will have a critical defect that may require surgery or other procedures in the first year of life.
Surgeons typically will start with a “valve repair and not put in any of this extra hardware because that, over time, could be more beneficial to the child,” Olson said. “But if that isn’t going to work, this is really the next option. This gives them the opportunity — in a case where there are no other options, we have this as a backup — that we can treat these patients and still give them a chance to live.”
2 MM DIFFERENCE
Before the 15-millimeter valve was developed, the only option for pediatric cardiac surgeons treating infants and small toddlers with heart defects requiring a mechanical-valve replacement was to implant a larger 17-millimeter valve.
“Although that doesn’t sound like a big difference, 2 millimeters, it makes all the difference in the world when you’re talking about an infant micro-valve because sometimes you just simply cannot fit in the 17,” said Jonathan Chen, co-director of the Seattle Children’s Hospital Heart Center and division chief for pediatric cardiothoracic surgery.
Chen helped lead the clinical trial that led to March 6 FDA approval of the 15-millimeter Masters Series Mechanical Heart Valve with Hemodynamic Plus.
The trial, which started in 2014, enrolled pediatric patients 5-years old or younger “who had a diseased, damaged or malfunctioning heart valve.” Prior to the trial, the valve had been available for emergency and compassionate-use cases through special petition to the FDA.
IMPLANT AT 9 DAYS OLD
The majority of 15-millimeter implants have taken place before a baby’s first birthday. The youngest infant to receive a valve was 9-days old.
Chen was the first physician in the trial to implant the 15-millimeter valve in a pediatric patient. That 2014 surgery saved the life of Sadie Rutenberg, now 3, said Lee’or Rutenberg, Sadie’s father, who lives in Seattle.
“When (he) came out of surgery and told us the surgery was a success — as a parent, it’s a moment I’ll never forget,” he said.
FDA approval means the 15-millimeter valves will be available on site when needed, even in emergencies, Chen said.
Before, if Chen decided that he needed to implant a 15-millimeter valve, he would have to get approval from the hospital’s Institutional Review Board and then order it and have it shipped to Seattle — a process that could take four or five days, he said.
“If you wanted to go on Monday, you’d have to wait until Friday for the operation, and the child is not getting any better on those interim days,” Chen said. “Now, we can just literally have it on the shelf. So if we decided we wanted to (implant one) today, we could just do it today, which is a huge advance for us.”
Another plus: the rotatable valve is designed for implantation in the mitral or aortic position, he said.
ORIGINS AT ST. JUDE
Legendary med-tech entrepreneur Manny Villafaña sketched the idea for a mechanical heart valve on the back of a napkin in October 1976 for St. Jude Medical, the company he founded. An enlarged version of the napkin hangs on the wall of the manufacturing facility in Little Canada.
“If you looked at this, compared to what is being made today, this is almost exactly what we’re doing,” Olson said. “There’s obviously been a lot of refinement over the years, but this is what started it all.”
The mechanical heart valves mimic the valve of a healthy heart, opening and closing with each heartbeat, permitting proper blood flow through the heart. They do not require batteries.
Abbott’s mechanical heart valves come in seven diameter sizes, ranging from 15- to 27-millimeter. The 15-millimeter valves are expected to remain in place for two to five years — until the child outgrows the valve, said Chad Green, principal research and development engineer for Abbott’s structural heart division. “As they grow, their body demands more blood flow.”
Patients will continue to receive a next size until they are able to receive an adult-sized valve.
Each 15-millimeter valve takes several weeks to manufacture; the entire valve is made in Little Canada. Parts of the larger valves are made at a sister plant in Puerto Rico.
‘SIMPLY THE RIGHT THING TO DO’
Abbott declined to disclose the sale price of the valve, saying only that it is priced similarly to its other mechanical valves. Specific procedural costs charged by the hospital are at the hospital’s discretion, Abbott officials said in a statement.
Abbott expects to produce about 200 valves a year. They represent less than 0.5 percent of the company’s total volume mix, Olson said.
“This is not a growth driver for Abbott,” he said. “By no means is the volume meaningful from the standpoint of a revenue projection — that’s not what this is about. We have this technology, and it’s simply the right thing to do. Why wouldn’t we provide this for the most needy — the children — of the world?”
Chen said Abbott should be applauded for their work.
“Kudos go to Abbott for pursuing a pediatric application because it is truly an orphan application,” he said.
“They’re not going to make billions of dollars off this. This is just out of the kindness of their hearts that they’re making this available to us.”